The U.S. Food & Drug Administration determined in 2015 that Partially Hydrogenated Oils (“PHOs”) are not “Generally Recognized as Safe,” or GRAS. PHOs are the leading source of trans fats in the human diet.
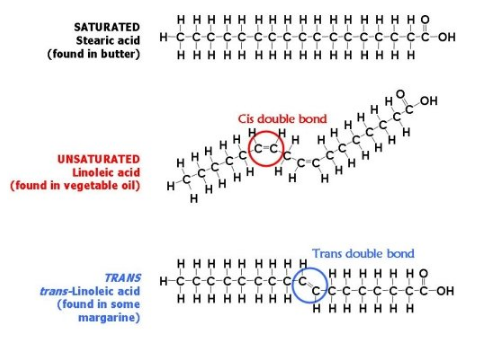
In the nutrition industry, much is disputed, but the unhealthy nature of trans fats, a form of unsaturated fat, is not. Trans fats are fats that have a unique chemical structure; they contain double bonds on opposite sides, whereas other unsaturated fats have double bonds on the same side, and saturated fats lack any double bonds.
Source: http://chemistrybasics.edublogs.org/2007/02/16/trans-fats/
Because of their unique structure, trans fats can pose significant health risks, such as heart disease and high cholesterol. While non-harmful trans fats do occur naturally in some foods, harmful artificial trans fats are created by pumping extra hydrogen molecules into oils, or “hydrogenating” the oil. In coming to its 2015 decision, the FDA reviewed extensive clinical data as well as public commentary, and found that eliminating PHOs from processed foods could save thousands of lives per year.
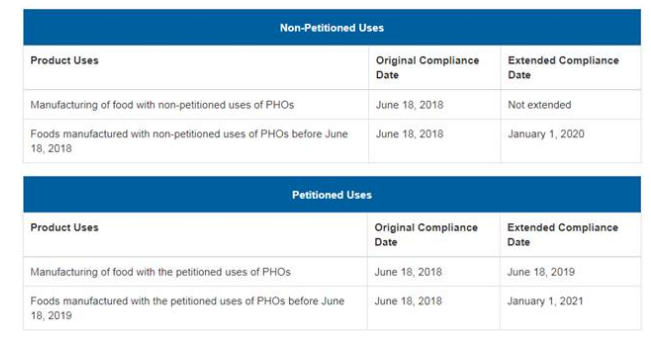
The FDA set June 18, 2018 as the deadline for compliance for all product uses, but on May 15, 2018 extended that deadline for all product uses except one. The new deadlines will occur in 2019, 2020, and 2021 as set forth below:
Source: https://www.fda.gov/Food/IngredientsPackagingLabeling/FoodAdditivesIngredients/ucm449162.htm
The FDA has stated that the extension is due largely to the 3-24 month shelf life of products containing PHOs such that these products will be in distribution for some time after the compliance date, as well as due to additional compliance requirements placed upon the food industry because of the FDA’s denial of a concurrent food additive petition by the Grocery Manufacturers Association.
For more, see the full notice by the FDA: https://s3.amazonaws.com/public-inspection.federalregister.gov/2018-10714.pdf